Focal Medical-Virginia Tech Press Release
May 4, 2023

Initiation of Canine Oral Cancer Study
Trial & Enrollment Details
Cary, NC and Roanoke, VA, May 04, 2023 — Focal Medical, Inc., a privately held, biopharmaceutical company developing novel therapeutic products based on its innovative local drug delivery technology platform today announced the initiation of a research study in collaboration with Virginia Tech’s Animal Cancer Care and Research Center.
The study will evaluate the anticancer activity of Focal Medical’s targeted iontophoretic technology platform against oral cancers in canine companion animals. The study is being conducted in collaboration with Dr. Ilektra Athanasiadi, DVM, Assistant Professor of Radiation Oncology in the Department of Small Animal Clinical Sciences at Virginia-Maryland College of Veterinary Medicine.
Focal Medical’s patented iontophoresis delivery system enables the internal, site-specific, active delivery of drugs directly and selectively to diseased target tissue or organs. The technology thus addresses the significant challenges and limitations of traditional (primarily oral and intravenous) systemic drug delivery approaches, including toxicity and first-pass metabolism.
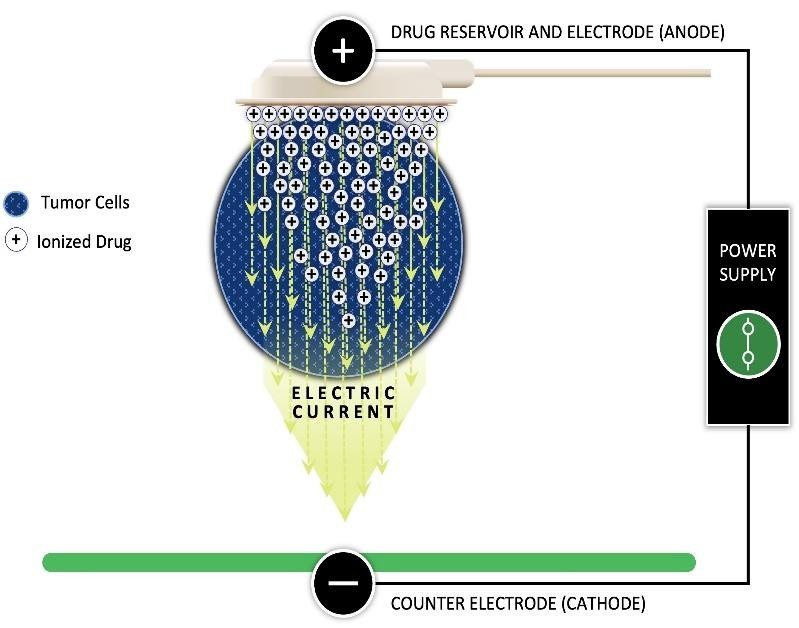
This study is treating oral cancer in canine companion animals using a single local carboplatin treatment directly to the tumor site. The key endpoints of this study are safety and feasibility of local carboplatin treatment, pharmacokinetic measurements, and evaluation of tumor volume following treatment. The study will be conducted at Virginia Tech’s Animal Cancer Care and Research Center in Roanoke, Virginia. Dogs diagnosed with either oral malignant melanoma or squamous cell carcinoma, weighing at least 33 lbs., and who are otherwise healthy are eligible for this study.
To inquire about enrollment, please contact Dr. Ilektra Athanasiadi:
oncology@vt.edu
(540) 526-2300
or visit the clinical trials website.
If this device works for head and neck cancers in dogs, the investigators hope it can be translated into human treatments in the future. Dr. Athanasiadi is excited about the potential to contribute to translational research. "The ability to deliver a therapeutic directly to the target, knowing that it potentially will work better and avoid systemic toxicity, that's amazing, especially when we deal with cancerous processes that are local.”
About Focal Medical
Focal Medical, Inc. is a privately held, biopharmaceutical company developing novel therapeutic products based on its innovative and patent protected local drug delivery technology platform. Focal Medical’s patented iontophoresis delivery system enables the internal, site-specific delivery of therapeutics actively, directly, and selectively to the diseased target tissue or organ. The technology thus addresses certain significant challenges and limitations of traditional systemic drug delivery approaches including toxicity and first-pass metabolism. Focal Medical’s lead product uses its patented iontophoresis system for the local delivery of gemcitabine (an FDA approved chemotherapeutic) actively and directly to the tumor to treat pancreatic cancer. Focal Medical is expanding its product focus into therapies for other solid tumors and genomic medicine products, all utilizing its innovative energy-based drug delivery system.
Focal Medical’s Board includes Joseph M. DeSimone Ph.D, Founder, the Sanjiv Sam Gambhir Professor of Translational Medicine and Chemical Engineering at Stanford University, Jen Jen Yeh, M.D. Founder, Professor, Departments of Surgery and Pharmacology, Vice Chair of Research Department of Surgery and Director of the Pancreatic Cancer Center of Excellence at the Lineberger Comprehensive Cancer Center, University of North Carolina School of Medicine and Paula Hammond, Institute Professor and Department Head Department of Chemical Engineering, Massachusetts Institute of Technology, along with executives, Michael Aldridge, CEO and Anthony Voiers, COO.
For More Information on Focal Medical:
www.focalmedical.co
Tony Voiers, COO
(919) 917-7324
tvoiers@focalmedical.co
Press Contact:
Andrew Mann
(540) 231-9005
andrewfjmann@vt.edu






